|
|
|
 |
 |
 |
 |
|
|
|
 |
|
|
КИЕЕРкЗс
|
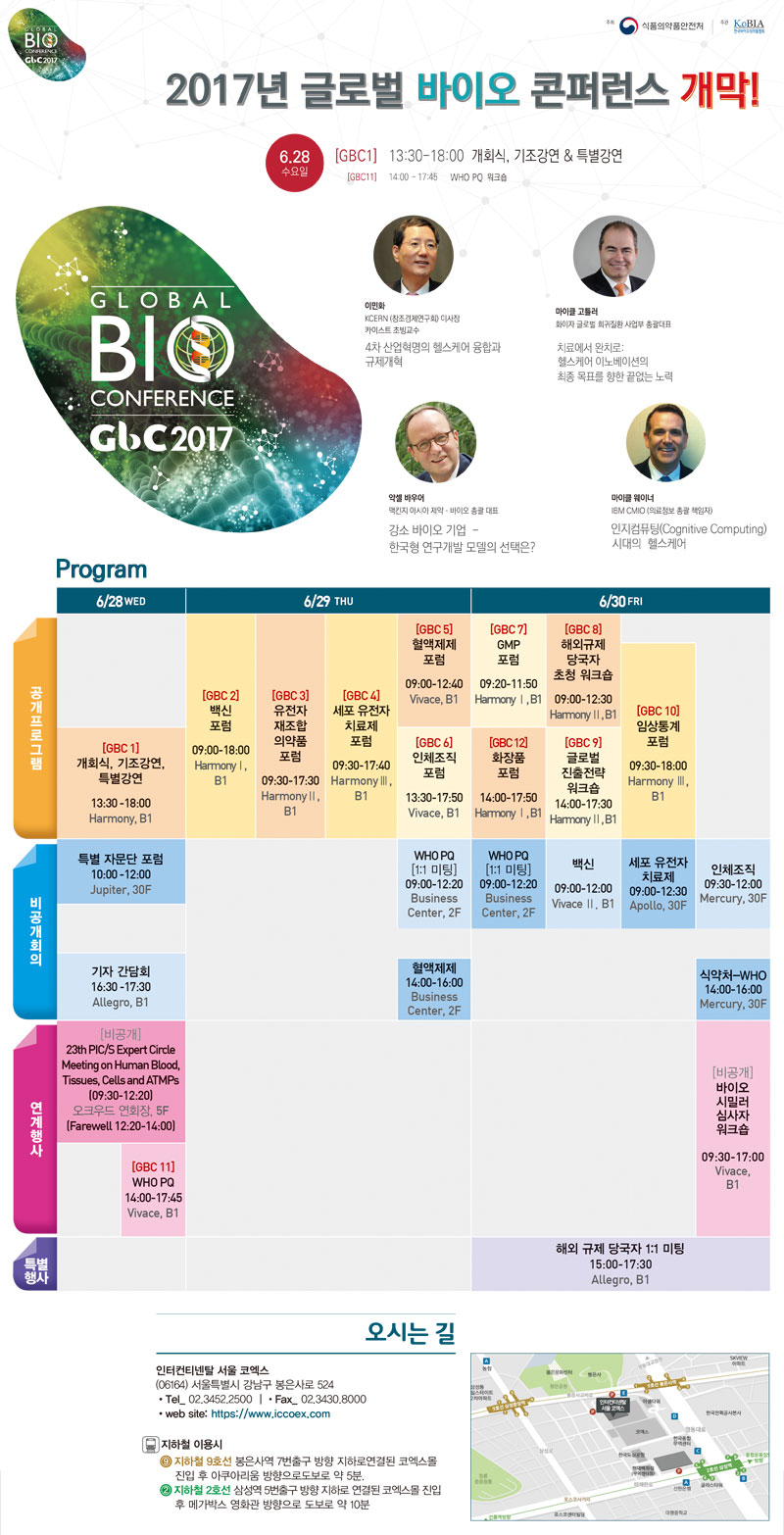
4Тї ЛъОїЧѕИэРЧ СпНЩ, ЙйРЬПРРЧОрЧА РЮНФСжАЃ ИТОЦ ДйОчЧб БЙСІЧрЛч АГУж
Ёр НФЧАРЧОрЧАОШРќУГ(УГРх МеЙЎБт)ДТ РЮЗљ АЧАПЁ СпУпРћ ПЊЧвРЛ ДуДчЧЯДТ ЙйРЬПРРЧОрЧАРЧ СпПфМКРЛ ОЫИЎАэ ЙйРЬПРРЧОрЧА ЛъОїРЛ ПьИЎГЊЖѓ ЙЬЗЁ ЧйНЩЛъОїРИЗЮ РАМКЧЯБт РЇЧб РќЗЋ ЕюРЛ АјРЏЧЯБт РЇЧЯПЉ ПРДТ 26РЯКЮХЭ 30РЯБюСіИІ ЁЎЙйРЬПРРЮНФ СжАЃЁЏРИЗЮ СіСЄЧЯАэ ДйОчЧб БЙСІЧрЛчИІ АГУжЧбДйАэ ЙрЧћДй.
Ёл РЬЙј СжАЃРК 4Тї ЛъОїЧѕИэРЧ СпНЩРИЗЮ КЮЛѓЧб ЙйРЬПРРЧОрЧАЛъОїРЧ СпПфМКПЁ ДыЧи АјАЈДыИІ ЧќМКЧЯАэ, АќЗУ ЛъОїРЛ РАМКЧЯБт РЇЧЯПЉ ПУЧи УГРН СіСЄЕЧОњРИИч, БЙГЛЁЄПм МЎЧаЕщРЛ УЪДыЧЯПЉ ЙйРЬПРРЧОрЧА АГЙпАњ АќЗУЧб УжНХ ЕПЧтАњ БдСІ СЄКИ ЕюРЛ АјРЏЧбДй.
Ёр РЬЙј ЁЎЙйРЬПРРЮНФ СжАЃЁЏ СжПфЧрЛчДТ ЁуРЧОрЧАНЧЛчЛѓШЃЧљЗТБтБИ(PIC/S) РќЙЎАЁКаАњ ШИРЧ ЁуWHOПЭ БЙГЛ СІОрЛч 1:1 ЙЬЦУ ЁуУЗДмЙйРЬПРРЧОрЧА ЦЏКАРкЙЎДм ЦїЗГ ЁуБлЗЮЙњЙйРЬПРФмЦлЗБНКРЬДй.
Ёл БлЗЮЙњЙйРЬПРФмЦлЗБНК(6.28-6.30)ПЁМДТ ЁЎ4Тї ЛъОїЧѕИэРЧ СпНЩ, ЙйРЬПРРЧОрЧАЁЏРЛ СжСІЗЮ ЙйРЬПРРЧОрЧА КаОп ММАшРћ БЧРЇРк, БдСІДчБЙРкЕщАњ ЧдВВ ПьИЎГЊЖѓ ЙйРЬПРРЧОрЧАРЛ ММАшРћ КъЗЃЕхЗЮ РАМКЧЯБт РЇЧб СіНФАњ АцЧшРЛ АјРЏЧбДй.
- ЦЏШї БтСЖАПЌПЁМДТ БлЗЮЙњСІОрАБЙ РАМКРќЗЋАњ ЙйРЬПРЧяНКФЩОю ЛѕЗЮПю ЦаЗЏДйРг ЕюПЁ ДыЧи ИЖРЬХЌ ПўРЬГЪ IBM ЧяНКФЩОю МіМЎРЧЗсСЄКИ УЅРгРк ЕюРЬ ЙпЧЅЧбДй.
- ЖЧЧб ЙщНХ, ММЦїЁЄРЏРќРкФЁЗсСІ, РЏРќРкРчСЖЧе Ею ЙйРЬПРРЧОрЧА КаОпКА ЦїЗГАњ GMP, РЮУМСЖСї ЦїЗГРЬ АГУжЕЧИч, ЦЏКАЧрЛчЗЮ БЙГЛ СІОрЛчАЁ МіУтРЛ ШёИСЧЯДТ СіПЊРЧ ЧиПм БдСІ ДчБЙРкПЭРЧ 1:1 ЙЬЦУРЛ СжМБЧбДй.......Дѕ КИБт
|
|
 |
|
КИЕЕРкЗс
|
НФОрУГ, ЙйРЬПРНУЙаЗЏ ЧуАЁНЩЛч КаОп ПЊЗЎ ДйНУ Чб Йј РдСѕ
- БЙСІРЧОрЧАБдСІРкЦїЗГ ЙйРЬПРНУЙаЗЏ ПіХЗБзЗь РЧРхБЙ ПЌРг -
Ёр НФЧАРЧОрЧАОШРќУГ(УГРх МеЙЎБт)ДТ ЁЎ2017Гт ЛѓЙнБт БЙСІРЧОрЧАБдСІРкЦїЗГ(IPRF) Йз БЙСІРЧОрЧАБдСІСЖШРЇПјШИ(ICH) УбШИЁЏПЁМ ПьИЎГЊЖѓАЁ IPRF ЙйРЬПРНУЙаЗЏ РЇХЗБзЗь РЧРхБЙРИЗЮ ПЌРгЕЧОњДйАэ ЙрЧћДй.
Ёи ЙйРЬПРНУЙаЗЏ ПіХЗБзЗь : ЙЬБЙ, РЏЗД Ею 11АГБЙ БдСІБтАќ Йз 3АГ БЙСІДмУМ МвМг 32Иэ БИМКЕЪ
Ёи ICH УбШИ: ФГГЊДй ИѓЦЎИЎПУ АГУж(5.28Ё6.1)
Ёл РЬЙј ПЌРгРИЗЮ ПьИЎГЊЖѓДТ ЙйРЬПРНУЙаЗЏ ЧуАЁЁЄНЩЛч КаОпПЁ ДыЧб ПЊЗЎРЛ БЙСІРћРИЗЮ ДйНУЧбЙј РЮСЄЙоОвРИИч, ЁЏ19ГтБюСі ЙйРЬПРНУЙаЗЏ КаОпРЧ БдСІСЖШ МБЕЕПЁ СпУпРћ ПЊЧвРЛ ДуДчЧЯАд ЕШДй.
Ёл ТќАэЗЮ IPRFДТ РЧОрЧА БдСІ АќЗУ СЄКИБГШЏ Йз ЛѓШЃЧљЗТРЛ РЇЧи БИМКЕШ БдСІДчБЙРкАЃ ШИРЧУМЗЮМ ЙйРЬПРНУЙаЗЏ, ММЦїФЁЗсСІ Ею 4АГ ПіХЗБзЗьРЛ ПюПЕЧЯАэ РжДй.
- ПьИЎГЊЖѓДТ ММАш УжУЪЗЮ ЧзУМ ЙйРЬПРНУЙаЗЏИІ ЧуАЁЧЯДТ Ею БдСІБтАќРЧ НХЗкМКРЛ РЮСЄЙоОЦ ЁЎ14Гт ICH КёШИПј БЙАЁЗЮДТ УГРНРИЗЮ ЙйРЬПРНУЙаЗЏ ПіХЗБзЗь РЧРхБЙРИЗЮ МБУтЕЧОњДй.......... Дѕ КИБт
|
 |
|
АГСЄАэНУ
|
ЁИЛ§ЙАЧаРћСІСІ ЕюРЧ ЧАИёЧуАЁЁЄНЩЛч БдСЄЁЙ РЯКЮАГСЄАэНУ
СжПф ГЛПы
АЁ. РЏРќРк БГСЄ БтМњ Ею АГЙпЕЧАэ РжДТ РЏРќРкФЁЗсСІРЧ ЦЏМКРЛ ЙнПЕЧб СЄРЧ АГМБ(ОШ СІ2СЖСІ15ШЃ)
ГЊ. ЙйРЬПРРЧОрЧА ЧуАЁНЩЛчРЧ НХМгМКАњ ПЙУјМК СІАэИІ РЇЧи ЧАИёЧуАЁ НЩЛч НУ БИКёРкЗс ПфАЧ ЙЬКёПЉКЮПЁ ДыЧб НХМг АЫХфИІ РЇЧб ПЙКёНЩЛч СІЕЕ ЕЕРд(ОШ СІ2СЖСІ25ШЃ, СІ38СЖСІ1Чз НХМГ)).......Дѕ КИБт
ЂК ЁИЛ§ЙАЧаРћСІСІ ЕюРЧ ЧАИёЧуАЁЁЄНЩЛч БдСЄЁЙ РќЙЎ КИЗЏАЁБт
|
 |
|
АГСЄАэНУ
|
ЁИШёБЭРЧОрЧА СіСЄПЁ АќЧб БдСЄЁЙ РЯКЮАГСЄАэНУ
СжПф ГЛПы
АЁ. ЕПСОСіЙцРЏЗЁ СпАЃПБСйБтММЦїИІ АГЙпДмАш ШёБЭРЧОрЧАРИЗЮ НХБд СіСЄЧЯАэ РЬЙЬ ШёБЭРЧОрЧАРИЗЮ СіСЄЧб ММИЎЦМДеРЧ ДыЛѓСњШЏРЛ КЏАцЧд.(ОШ КАЧЅ 1 Йз КАЧЅ 2)
1) РћПыДыЛѓРЬ ЕхЙААэ РћР§Чб ДыУМРЧОрЧАРЬ ОјОю БфБоЧб ЕЕРдРЬ ЧЪПфЧб РЧОрЧАРЛ ШёБЭРЧОрЧАРИЗЮ УпАЁ СіСЄЧв ЧЪПфАЁ РжРН.
2) ЕПСОСіЙцРЏЗЁ СпАЃПБСйБтММЦїИІ АГЙпДмАш ШёБЭРЧОрЧАРИЗЮ ЛѕЗгАд СіСЄЧЯАэ, РЬЙЬ ШёБЭРЧОрЧАРИЗЮ СіСЄЧб ММИЎЦМДеРЧ ДыЛѓСњШЏРЛ КЏАцЧд.
3) ШёБЭРЧОрЧА СіСЄРЛ ШЎДыЧдРИЗЮНс ШёБЭСњШЏ ШЏРкРЧ ФЁЗсСІ МБХУ БтШИАЁ ДѕПэ ФПСњ АЭРИЗЮ БтДыЕЪ. ........Дѕ КИБт
|
 |
|
РЧАпСЖШИ
|
РЧПјЙпРЧ ЁИБЙАЁЧЪМіРЧОрЧАРЧ АјБо Йз АќИЎПЁ АќЧб Й§ЗќЁЙСІСЄ(ОШ) РЧАпСЖШИ (БЧЙЬЧѕ РЧПј ДыЧЅЙпРЧ)
СжПф ГЛПы
БЙАЁЧЪМіРЧОрЧААќИЎСОЧеАшШЙРЧ МіИГ Ею БЙАЁЧЪМіРЧОрЧААќИЎПЁ АќЧб СпПфЧб ЛчЧзРЛ НЩРЧЧЯБт РЇЧЯПЉ БЙЙЋУбИЎ МвМгРИЗЮ БЙАЁЧЪМіРЧ ОрЧААќИЎРЇПјШИИІ ЕЮАэ, РЬПЭ АќЗУЧб ПЌБИЛчОї, ХыАшСЖЛчЛчОї Йз СЄКИЛчОїРЛ НУЧрЧд. КИАЧКЙСіКЮРхАќМвМгРИЗЮ АјАјСІОрЛчИІ МГИГЧЯАэ АјАјСІОрЛчДТ БЙАЁЧЪМіРЧОрЧАРЧ АјБоАњ АќИЎ ЕюРЧ ЛчОїРЛ МіЧрЧЯЕЕЗЯ Чд
РЧАп СІУт БтЧб: '17. 06. 28(Мі)
СІУт ОчНФРК ЧљШИ ШЈЦфРЬСіПЁМ ШЎРЮЧЯНУБт ЙйЖјДЯДй........Дѕ КИБт
|
 |
|
УЄПыАјАэ
|
НФЧАРЧОрЧАОШРќУГ (НФЧАРЧОрЧАОШРќЦђАЁПј) '17Гт СІ2ШИ НЩЛчАќ УЄПы
АјАэИэ: НФЧАРЧОрЧАОШРќЦђАЁПј НЩЛчАќ УЄПы
СЂМіБтАЃ: '17.6.22.(Иё) ~ 7.7.(Бн) 18:00
СЂМіЙцЙ§: НФОрУГ ПьМіРЮРчУЄПыНУНКХл ПТЖѓРЮ СЂМі.......ЙйЗЮ АЁБт
|
|
 |
|
 |
| НХУЛРк |
НТРЮРЯ |
СІЧАИэ |
РгЛѓ |
ДмАш |
| МПяДыЧаБГКДПј |
20170623 |
РчЙпМК / РќРЬМК ЕЮАцКЮ ЦэЦђЛѓЧЧОЯ ШЏРкИІ ДыЛѓРИЗЮ Чб ДѕЙпЗчИЪАњ ДмРЯФЁЗс ПфЙ§Ањ ДѕЙпЗчИЪ ЦЎЗЙИсИЎЙТИП КДПы ФЁЗсПфЙ§ПЁ ДыЧб СІ2Лѓ РгЛѓНУЧш |
2Лѓ |
MEDI4736,
Tremelimumab |
|
РЯОчОрЧА(Сж)
|
20170621 |
АЧАЧб ПЕРЏОЦ Йз МвОЦ, УЛМвГтРЛ ДыЛѓРИЗЮ 'РЯОч РЮЧУЗчПЃРк КаЧвЙщНХ 4АЁ'РЧ ИщПЊПјМК Йз ОШРќМКРЛ ЦђАЁЧЯБт РЇЧб АјАГ(Part1), ДмРЯБК(Part1), ЙЋРлРЇЙшСЄ(Part2,Part3), РЬСпДЋАЁИВ(Part2,Part3), ШАМКДыСЖ(Part2,Part3) СІ3Лѓ РгЛѓНУЧш |
3Лѓ |
РЯОчЧУЗчЙщНХ4АЁСж
(РЮЧУЗчПЃРкКаЧвЙщНХ) |
| Ф§ХИРЯСюЦЎЗЃНКГЛМХГЮФкИЎОЦ(Сж) |
20170620 |
РчЙпМК ЖЧДТ РќРЬМК ЕЮАцКЮ ЦэЦђЛѓЧЧММЦїОЯ НУЧшДыЛѓРкРЧ ФЁЗсИІ РЇЧб ЦшКъЗбИЎСжИПАњ КДПыЧб ХЛИЎИ№Ае ЖѓЧуЦФЗОКЄРЧ СІ 1b/3Лѓ ДйБтАќ, ЙЋРлРЇ ЙшСЄ РгЛѓНУЧш |
1/3Лѓ |
ХЛИЎИ№Ае ЖѓЧуЦФЗОКЄ (Talimogene Laherparepvec) |
| (Сж)ОЫХзПРСЈ |
20170616 |
HER2 ОчМК РЏЙцОЯ ДыЛѓРк Сп ЧЅСиФЁЗс НЧЦаЧб РќРЬИІ АЁСј СјЧрМК ЖЧДТ РчЙпМК ШЏРкИІ ДыЛѓРИЗЮ ALT-P7РЧ ОШРќМК, ГЛОрМК Йз ОрЕПЧаРЛ ЦђАЁЧЯБт РЇЧб АјАГ, ДмАшРћ СѕЗЎ, СІ 1Лѓ РгЛѓНУЧш |
1Лѓ |
ALT-P7 |
|
|
|
|
|
 |
|
|
|
| FDA |
|
Drug Name and
FDA Appl. # |
Active Ingredients |
Submission
Classification |
Company |
Approval Date |
COTEMPLA XR-ODT
NDA #205489 |
METHYLPHENIDATE |
НХПыЗЎ |
NEOS THERAP INC |
06/19/2017 |
MYDAYIS
NDA #022063 |
MIXED SALTS OF A SINGLE-ENTITY AMPHETAMINE |
НХПыЗЎ |
SHIRE DEV LLC |
06/20/2017 |
RITUXAN HYCELA
BLA #761064 |
HYALURONIDASE
RITUXIMAB |
- |
GENENTECH INC |
06/22/2017 |
BEVYXXA
NDA #208383 |
BETRIXABAN |
НХЙАСњ |
PORTOLA PHARMA INC |
06/23/2017 |
|
|
 |
|
| EMA |
|
| Name |
Active Substance |
Therapeutic areaCompany |
Date of authorisation
/refusal |
| Spinraza |
nusinersen sodium |
Muscular Atrophy, Spinal |
30/05/2017 |
| Brineura |
cerliponase alfa |
Neuronal Ceroid-Lipofuscinoses |
30/05/2017 |
| Elmiron |
pentosan polysulfate sodium |
Cystitis, Interstitial |
02/06/2017 |
|
|
|
 |
|
|
|
|
| Clinical.gov ЙЬБЙ |
NCT
Number |
Title |
Conditions |
Interventions |
Sponsor
/Collaborators |
Phases |
| NCT03197506 |
Pembrolizumab and Standard Therapy in Treating Patients With Glioblastoma |
Glioblastoma
Gliosarcoma
Supratentorial Glioblastoma
|
Radiation: External Beam Radiation Therapy
Other: Laboratory Biomarker Analysis
Biological: Pembrolizumab
Radiation: Radiation Therapy
Drug: Temozolomide
Procedure: Therapeutic
Conventional Surgery
|
Mayo Clinic
National Cancer Institute (NCI) |
Phase 2 |
| NCT03197025 |
Immunotherapy With
E6 T Cell Receptor (TCR)
T Cells for Vulvar High-Grade Squamous Intraepithelial Lesions |
Human Papillomavirus
HPV-16
High Grade Squamous Intraepithelial Lesion |
Drug: Aldesleukin
Biological: E6 TCR |
Memorial Sloan Kettering Cancer Center
Y-Mabs, Inc |
Phase 1 |
| NCT03196232 |
Epacadostat and Pembrolizumab in Treating Patients With Metastatic or Unresectable Gastroesophageal Junction or Gastric Cancer |
Gastric Adenocarcinoma
Gastroesophageal Junction Adenocarcinoma
Recurrent Esophageal Carcinoma
Recurrent Gastric Carcinoma
Stage IV Esophageal Cancer AJCC v7
Stage IV Gastric Cancer AJCC v7
Unresectable Esophageal Carcinoma |
Drug: Epacadostat
Other: Laboratory
Biomarker Analysis
Biological: Pembrolizumab
|
Pamela L. Kunz
Stanford University |
Phase 2 |
| NCT03196401 |
A Study of Immunotherapy Plus Radiation Therapy to Stimulate Immunity in Solitary Bone Plasmacytoma |
Solitary Bone Plasmacytoma |
Biological: Peri-urethral and clitoral injections |
Center for Vulvovaginal Disorders |
Phase 2 |
| NCT03197935 |
A Study to Investigate Atezolizumab and Chemotherapy Compared With Placebo and Chemotherapy in the Neoadjuvant Setting in Participants With Early Stage Triple Negative Breast Cancer |
Triple-negative
Breast Cancer |
Drug: Atezolizumab (MPDL3280A),
an engineered anti-PDL1 antibody
Drug: Placebo
Drug: Nab-paclitaxel
Drug: Doxorubicin
Drug: Cyclophosphamide
Drug: Filgrastim
Drug: Pegfilgrastim |
Hoffmann-La Roche |
Phase 3 |
|
|
 |
|
| Clinical.gov РЏЗД |
NCT
Number |
Title |
Conditions |
Interventions |
Sponsor
/Collaborators |
Phases |
| NCT03197467 |
Neoadjuvant Anti PD-1 Immunotherapy in Resectable Non-small Cell Lung Cancer |
Non-small Cell Lung Cancer (NSCLC) |
Drug: Pembrolizumab |
AIO-Studien-gGmbH|Merck Sharp & Dohme Corp. |
Phase 2 |
| NCT03197935 |
A Study to Investigate Atezolizumab and Chemotherapy Compared With Placebo and Chemotherapy in the Neoadjuvant Setting in Participants With Early Stage Triple Negative Breast Cancer |
Triple-negative Breast Cancer |
Drug: Atezolizumab (MPDL3280A),
an engineered anti-PDL1 antibody
Drug: Placebo
Drug: Nab-paclitaxel
Drug: Doxorubicin
Drug: Cyclophosphamide
Drug: Filgrastim
Drug: Pegfilgrastim |
Hoffmann-La Roche |
Phase 3 |
|
|
 |
|
|
| Clinical.gov СпБЙ |
NCT
Number |
Title |
Conditions |
Interventions |
Sponsor
/Collaborators |
Phases |
|
NCT03198052
|
PSCA-CAR-T or MUC1-CAR-T for Cancer With PSCA/MUC1 Expression |
Lung Cancer
Cancer
Immunotherapy
CAR-T Cell |
Genetic: PSCA or MUC1 targeting CAR-T cells |
Second Affiliated Hospital of Guangzhou Medical University |
Phase 1 |
| NCT03196986 |
MIL60 Versus Bevacizumab in Patients With Treatment-naïve Non-squamous Non-small Cell Lung Cancer |
Non-small Cell Lung Cancer |
Drug: MIL60
Drug: Bevacizumab |
Beijing Mabworks Biotech Co., Ltd. |
Phase 3 |
| NCT03196830 |
CAR-T for R/R B-NHL |
Relapsed Non Hodgkin Lymphoma
Refractory Non-Hodgkin Lymphoma
CAR - T CD19/CD20/CD22/CD30 |
Biological: CAR-T |
The First Affiliated Hospital of Soochow University
Shanghai Unicar-Therapy Bio-medicine Technology Co.,Ltd |
Phase 2 |
| NCT03195491 |
A Study of Non-Small Cell Lung Cancer (NSCLC) Patients Receiving Second-Line Nivolumab Monotherapy in Asia |
Lung Cancer
Non-Small Cell Lung Cancer |
Biological: Nivolumab |
Bristol-Myers Squibb |
Phase 3 |
| NCT03195478 |
Study of Nivolumab in Combination With Ipilimumab in Chinese Subjects With Previously Treated Advanced or Recurrent Solid Tumors |
Solid Tumor |
Drug: Nivolumab
Drug: Ipilimumab |
Bristol-Myers Squibb |
Phase 1 |
|
|
 |
|
| Clinical.gov РЯКЛ |
NCT
Number |
Title |
Conditions |
Interventions |
Sponsor
/Collaborators |
Phases |
|
NCT03197935
|
A Study to Investigate Atezolizumab and Chemotherapy Compared With Placebo and Chemotherapy in the Neoadjuvant Setting in Participants With Early Stage Triple Negative Breast Cancer |
Triple-negative Breast Cancer |
Drug: Atezolizumab (MPDL3280A), an engineered anti-PDL1 antibody
Drug: Placebo
Drug: Nab-paclitaxel
Drug: Doxorubicin
Drug: Cyclophosphamide
Drug: Filgrastim
Drug: Pegfilgrastim |
Hoffmann-La Roche |
Phase 3 |
|
|
|
|
 |
|
|
|
| ЙйРЬПРITЧУЗЇЦћ '17Гт НХБдСІАјСЄКИ ОШГЛ |
| ЙйРЬПРITЧУЗЇЦћРЧ 2017Гт НХБд СІАј СЄКИ |
|
2017ГтЕЕПЁДТ СіГ Чи БдСІЁЄЛъОїСЄКИИІ СІАјЧЯПДДј 12АГБЙ ПмПЁ КЃЦЎГВ, РЮЕЕГзНУОЦ, ЦФХАНКХКРЧ 3АГБЙ СЄКИИІ УпАЁЗЮ СІАјЧв ПЙСЄРдДЯДй. ИЙРК АќНЩАњ РЬПыРЛ КЮХЙ ЕхИГДЯДй.
|
 |
| ЙйРЬПРITЧУЗЇЦћ РќЙЎФСМГЦУ ОШГЛ |
|
2017ГтПЁДТ СжПф БЧПЊ(ЙЬБЙ, РЏЗД, СпБЙ)КА РќЙЎ ФСМГЦУ ОїУМРЧ РЮЧуАЁ СОЧе СіПј МКёНК(2017 УЗДмЙйРЬПРРЧОрЧА ЧиПмСјУт СОЧеСіПј ЛчОї ЁКЙйРЬПРITЧУЗЇЦћЁЛ РќЙЎФСМГЦУ)ИІ ЛѕЗгАд НЧНУЧв ПЙСЄРдДЯДй. ИЙРК НХУЛ ЙйЖјДЯДй.
|
| 'РќЙЎФСМГЦУ' РЬЖѕ? |
ЙйРЬПРРЧОрЧА ЧиПм РЮЧуАЁ АќЗУ ОїЙЋАцЧшРЬ РжДТ РќЙЎ ФСМГЦУ БтАќРЧ
1:1(ФСМГЦУБтАќ:ФСМГЦУ РЧЗк ОїУМ) ФСМГЦУ
|
|
 |
|
ШёИСЧЯНУДТ ОїУМВВМДТ ЧљШИ ЖЧДТ ЙйРЬПРITЧУЗЇЦћ ШЈЦфРЬСіРЧ ЁЎЙйРЬПРITЧУЗЇЦћ РќЙЎФСМГЦУ НХУЛЁЄСЂМі АјАэЁЏИІ ШЎРЮЧЯНУАэ,
ЁЎЙйРЬПРРЧОрЧА ЧиПмСјУт РќЙЎФСМГЦУ РЧЗкМЁЏИІ РлМКЧЯНУОю РЬИоРЯ(bpis@kobia.kr) НХУЛЁЄСЂМіЧЯНУБт ЙйЖјДЯДй.
ЕПРЯ БтОїРК УжДы 5АЧБюСі НЩШ, ПЌАш ФСМГЦУ(ДмАшКА) РЧЗкАЁ АЁДЩЧеДЯДй.
|
|
| ЂК ЙйРЬПРITЧУЗЇЦћ РќЙЎФСМГЦУ СЂМіАјАэ ЙйЗЮАЁБт |
|
| ЂК ЙйРЬПРITЧУЗЇЦћ ЙйЗЮАЁБт |
 |
|
|
|
 |
|
|
|
|
|
 |
|
|
|
|
|
 > СЄКИИЖДч > E-ДКНКЗЙХЭ > РќУМ
> СЄКИИЖДч > E-ДКНКЗЙХЭ > РќУМ
 > СЄКИИЖДч > E-ДКНКЗЙХЭ > РќУМ
> СЄКИИЖДч > E-ДКНКЗЙХЭ > РќУМ