Untitled Document
 |
 |
| |
|
 |
 |
 |
 |
| Ёс НФЧАРЧОрЧАОШРќУГ МвНФ |
 |
| ИХДКОѓ/СіФЇ |
ЁИ3D ЙйРЬПРЧСИАЦУ СІЧА Ею ММЦї-СіСіУМ КЙЧеСІЧАРЧ ЦђАЁ АЁРЬЕхЖѓРЮЁЙ Йз
ЁИРЏРќРкФЁЗсСІ КёРгЛѓНУЧш ЦђАЁ АЁРЬЕхЖѓРЮЁЙ ЙпАЃ |
 |
НФЧАРЧОрЧАОШРќУГ(УГРх ЗљПЕСј) НФЧАРЧОрЧАОШРќЦђАЁПјРК 3D ЙйРЬПРЧСИАЦУ СІЧА АГЙпПЁ ЧЪПфЧб СжПф АэЗСЛчЧзРЛ ДуРК ЁЎ3D ЙйРЬПРЧСИАЦУ СІЧА Ею ММЦї-СіСіУМ КЙЧеСІЧАРЧ ЦђАЁ АЁРЬЕхЖѓРЮЁЏРЛ СІСЄЁЄЙпАЃЧпДйАэ ЙрЧћДй.
СжПф ГЛПыРК Ёу3D ЙйРЬПРЧСИАЦУ СІЧА Ею ММЦї-СіСіУМ КЙЧеСІЧАРЧ КаЗљ БтСи Йз СЄРЧ ЁуЧАСњЁЄЦЏМККаМЎ РкЗс ЙќРЇ Йз АэЗСЛчЧз ЁуКёРгЛѓ ОШРќМКЁЄРЏШПМК ЦђАЁНУ АэЗСЛчЧз ЁуРгЛѓНУЧш НУ АэЗСЛчЧз ЕюРЬДй.
ЖЧЧб РЏРќРкСЖРл БтМњЗЮ РЏРќРкФЁЗсСІИІ АГЙпЧЯДТ АцПь ЧЪПфЧб СжПф АэЗСЛчЧзРЛ ДуРК ЁЎРЏРќРкФЁЗсСІ КёРгЛѓНУЧш ЦђАЁ АЁРЬЕхЖѓРЮЁЏРЛ АГСЄЁЄЙпАЃЧпДй.
СжПф АГСЄ ГЛПыРК ЁуРЏРќРкФЁЗсСІ АГЙп НУ ЕЖМКПЁ АќЧб РкЗс ЁуРЏРќРкФЁЗсСІ АГЙп НУ ОрИЎРлПыПЁ АќЧб РкЗс ЕюРЬДй. |
|
 |
| ЙЮПјРЮОШГЛМ |
УЗДмЙйРЬПРРЧОрЧА БдСІАњЧа ЛѓДу ЛчЗЪС§[ЙЮПјРЮ ОШГЛМ] |
 |
| ММЦїФЁЗсСІ Йз РЏРќРкФЁЗсСІ НЩЛч АќЗУ РкСжЙЏДТ СњЙЎАњ ДфКЏРЛ И№РК ЛѓДуЛчЗЪС§ |
|
 |
| АјСі/АјАэ |
2017Гт РЧОрЧАЧуАЁЦЏЧуПЌАшСІЕЕ ПЕЧтЦђАЁ АсАњКИАэМ |
 |
ОрЛчЙ§ СІ50СЖРЧ11ПЁ ЕћЖѓ ЦЧИХБнСі, ПьМБЦЧИХЧАИёЧуАЁ ЕюРЬ БЙГЛ СІОрЛъОї, КИАЧСЄУЅ, АэПыСѕАЈ ЕюПЁ ЙЬФЁДТ ПЕЧтРЛ
КаМЎ, ЦђАЁЧб КИАэМИІ КйРгАњ ААРЬ АјАГЧеДЯДй.
* (ПЌБИБтАќ) МПяДыЧаБГ ЛъЧаЧљЗТДм(ПЌБИУЅРгРк : РЬХТСј БГМі) |
|
 |
| АјСі/АјАэ |
WHO РЮЧУЗчПЃРк ЙщНХ, EЧќ АЃПА ЙщНХ, ЙйРЬПРНУЙаЗЏ ЕюПЁ АќЧб АЫХфРЧАп МіЗХ('18.1.18БюСі) |
 |
| НФЧАРЧОрЧАОШРќУГДТ ММАшКИАЧБтБИ(WHO) ЙйРЬПРРЧОрЧА ЧЅСиШ КаОп ЧљЗТМОХЭЗЮМ БЙСІ БтСи СІ,АГСЄПЁ АјЕПРИЗЮ ТќПЉЧЯАэ РжНРДЯДй. РЬПЭ АќЗУЧЯПЉ WHOПЁМ ОЦЗЁПЭ ААРЬ РЮЧУЗчПЃРкЙщНХ, EЧќ АЃПАЙщНХ Йз ЙйРЬПРНУЙаЗЏРЧ АЁРЬЕхЖѓРЮ(ОШ)ПЁ ДыЧб АЫХфРЧАпРЛ МіЗХЧЯАэ РжНРДЯДй.
АЁ. РЮЧУЗчПЃРк ЙщНХ ШФКИ ЙйРЬЗЏНКРЧ ЧАСњАќИЎПЁ АќЧб Л§ЙАОШРќ РЇЧшЦђАЁ Йз АЁРЬЕхЖѓРЮ
ГЊ. РчСЖЧе EЧќ АЃПА ЙщНХРЧ ЧАСњ, ОШСЄМК Йз ШПДЩ ШЎРЮРЛ РЇЧб БЧРхЛчЧз
Дй. WHO СњРЧ Йз РРДфС§: ЙйРЬПРНУЙаЗЏ СІЧА
АЂ АЁРЬЕхЖѓРЮ АЁ~ДйПЁ ДыЧб АЫХфРЧАпРЬ РжДТ АцПь, 2018.1.18.(Иё)БюСі АЂ УЗКЮЕШ РЧАпОчНФМИІ РлМКЧЯПЉ ЧљШИ(johs8739@kobia.kr)ЗЮ СІУтЧЯПЉ СжНУБт ЙйЖјДЯДй. |
|
 |
АјЙЋПј
СіФЇМ |
Л§ЙАРЧОрЧА Йз ЧбОр(Л§Ор)СІСІ РЇЧиМК АќИЎАшШЙ НЩЛч Йз УГИЎ Р§Тї(АјЙЋПј СіФЇМ) |
 |
| Л§ЙАРЧОрЧА Йз ЧбОр(Л§Ор)СІСІ РЇЧиМК АќИЎАшШЙ НЩЛч Йз УГИЎ Р§Тї(АјЙЋПј СіФЇМ) (rev1) |
|
 |
АјЙЋПј
СіФЇМ |
ЙйРЬПРЛ§ОрБЙ МвАќ ЧіРхСЁАЫ СіФЇ |
 |
| ЙйРЬПРРЧОрЧА, ЧбОр(Л§Ор)СІСІ/ЧбОрРч, ШРхЧА, РЧОрПмЧА, РЮУМСЖСї - (АјЙЋПј СіФЇМ) |
|
 |
| |
|
| Ёс БЙГЛ ЙйРЬПРРЧОрЧА РгЛѓНУЧш |
 |
| НХУЛРк |
НТРЮРЯ |
СІЧАИэ |
НУЧш
СІИё |
ДмАш |
ЧбБЙ
ПЅПЁНКЕ№(РЏ) |
20171218 |
РЬРќПЁ ФЁЗсЙоРК РЬЗТРЬ ОјРИИч СООчПЁМ PD-L1РЬ ОчМК(TPS ЁУ 50%)РЮ СІIVБт РќРЬМК КёМвММЦї ЦѓОЯ ШЏРкИІ ДыЛѓРИЗЮ Pembrolizumab + IpilimumabРЛ Pembrolizumab + РЇОрАњ КёБГ ЦђАЁЧЯДТ СІ3Лѓ, ЙЋРлРЇЙшСЄ, РЬСпДЋАЁИВ РгЛѓНУЧш(KEYNOTE-598) |
3Лѓ |
MK-3475 |
СжНФШИЛч
ПЁНКПЄЙйРЬСЈ |
20171218 |
НХРхРЬНФ МіЧ§РкПЁМ CMVПЭ BKV РчШАМКШ ПЙЙц DNA ЙщНХРЮ BD03 БйРАХѕПЉРЧ ГЛОрМКАњ ОШРќМК ЦђАЁИІ РЇЧб РќЧтРћ, АјАГ, ПыЗЎСѕЗЎ, ДйБтАќ, 1Лѓ РгЛѓНУЧш |
1Лѓ |
BD03 |
|
 |
| Ёс ЧиПм РгЛѓ ЧіШВ |
 |
| Ёр ЙЬБЙ |
|
| NCT Number |
Title |
Conditions |
Interventions |
Sponsor
Collaborators |
Phases |
| NCT03387098 |
QUILT-3.070: NANT Pancreatic Cancer Vaccine: Molecularly Informed Integrated Immunotherapy in Subjects With Pancreatic Cancer Who Have Progressed on or After Standard-of-care Therapy |
Pancreatic Cancer |
Drug: Aldoxorubicin HCl|Biological: ALT-803|Biological: ETBX-011|Biological: GI-4000|Biological: haNK for infusion|Biological: avelumab|Biological: bevacizumab
...... |
NantKwest, Inc. |
Phase 1
Phase 2 |
| NCT03368729 |
Niraparib in Combination With Trastuzumab in Metastatic HER2+ Breast Cancer |
Metastatic Breast Cancer|HER2 Positive Breast Carcinoma |
Drug: Niraparib|Drug: Trastuzumab |
University of Alabama at Birmingham
Translational Breast Cancer Research Consortium| |
Phase 1
Phase 2 |
| NCT03088878 |
A Study of the Cirmtuzumab and Ibrutinib in Patients With B-Cell Lymphoid Malignancies |
B-cell Chronic Lymphocytic Leukemia|Small Lymphocytic Lymphoma|Mantle Cell Lymphoma |
Drug: Cirmtuzumab|Drug: Ibrutinib|Drug: Cirmtuzumab plus ibrutinib |
University of California, San Diego
Oncternal Therapeutics, Inc |
Phase 1
Phase 2 |
| NCT03368859 |
A Study of ABT-165 Plus FOLFIRI vs Bevacizumab Plus FOLFIRI in Subjects With Metastatic Colorectal Cancer Previously Treated With Fluoropyrimidine/Oxaliplatin and Bevacizumab |
Cancer - Metastatic Colorectal |
Drug: ABT-165|Drug: Bevacizumab|Drug: Irinotecan|Drug: Leucovorin|Drug: Fluorouracil - bolus|Drug: Fluorouracil - infusion |
AbbVie |
Phase 2 |
| NCT03367715 |
Nivolumab, Ipilimumab, and Short-course Radiotherapy in Adults With Newly Diagnosed, MGMT Unmethylated Glioblastoma |
MGMT-unmethylated Glioblastoma (GBM)|GBM |
Drug: Nivolumab|Drug: Ipilimumab|Radiation: Radiation Therapy (RT) |
New York University School of Medicine |
Phase 2 |
| NCT03272633 |
Irradiated Donor Cells Following Stem Cell Transplant in Controlling Cancer in Patients With Hematologic Malignancies |
Acute Lymphoblastic Leukemia|Acute Myeloid Leukemia in Remission|Hematopoietic Cell Transplantation Recipient|JAK2 Gene Mutation|Loss of Chromosome 17p|Mantle Cell Lymphoma|Minimal Residual Disease..... |
Procedure: Allogeneic Hematopoietic Stem Cell Transplantation|Biological: Irradiated Allogeneic Cells... |
Rutgers, The State University of New Jersey|National Cancer Institute (NCI) |
Phase 2 |
| NCT03358706 |
A Study to Evaluate the Effect of Ustekinumab on Cytochrome P450 Enzyme Activities Following Induction and Maintenance Dosing in Participants With Moderate to Severe Crohn's Disease |
Crohn Disease |
Drug: Ustekinumab IV Infusion|Drug: Ustekinumab SC Injection|Drug: Midazolam 2 mg|Drug: Warfarin 10 mg|Drug: Vitamin K 10 mg|Drug: Omeprazole 20 mg|Drug: Dextromethorphan 30 mg|Drug: Caffeine 100 mg |
Janssen Research & Development, LLC |
Phase 1 |
|
|
 |
| Ёр РЏЗД |
|
|
| NCT Number |
Title |
Conditions |
Interventions |
Sponsor
/Collaborators |
Phases |
КёАэ |
| NCT03295383 |
Randomized Clinical Trial Comparing the Safety and Efficacy of Rituximab Versus Oral Cyclophosphamide in Severe Forms of Mucous Membrane Pemphigoid |
Severe Forms of Mucous Membrane Pemphigoid |
Drug: Rituximab 1g IV|Drug: Cyclophosphamide 50Mg Oral Tablet|Drug: Placebo of Rituximab|Drug: Placebo Oral Tablet |
University Hospital, Rouen |
Phase 3 |
ЧСЖћНК |
| NCT03387046 |
A Pilot Study in Subjects With Relapsing Remitting Multiple Sclerosis (RR-MS) |
Multiple Sclerosis, Relapsing-Remitting |
Dietary Supplement: D-aspartate
Drug: Placebo
Biological: IFN beta-1a
Drug: Methylprednisolone |
Merck KGaA |
Phase 2 |
ЕЖРЯ |
| NCT03358706 |
A Study to Evaluate the Effect of Ustekinumab on Cytochrome P450 Enzyme Activities Following Induction and Maintenance Dosing in Participants With Moderate to Severe Crohn's Disease |
Crohn Disease |
Drug: Ustekinumab IV Infusion|Drug: Ustekinumab SC Injection|Drug: Midazolam 2 mg|Drug: Warfarin 10 mg|Drug: Vitamin K 10 mg|Drug: Omeprazole 20 mg|Drug: Dextromethorphan 30 mg|Drug: Caffeine 100 mg |
Janssen Research & Development, LLC |
Phase 1 |
ЕЖРЯ |
|
|
 |
| Ёр СпБЙ |
|
| NCT Number |
Title |
Conditions |
Interventions |
Sponsor
/Collaborators |
Phases |
| NCT03380689 |
Phase I Study of Biweekly SIRB Regimen for Metastatic Colorectal Cancer |
Metastatic Colorectal Cancer |
Drug: S-1|Drug: Irinotecan|Drug: Bevacizumab |
Fujian Cancer Hospital |
Phase 1 |
|
|
 |
| Ёс ЧљШИ МвНФ |
 |
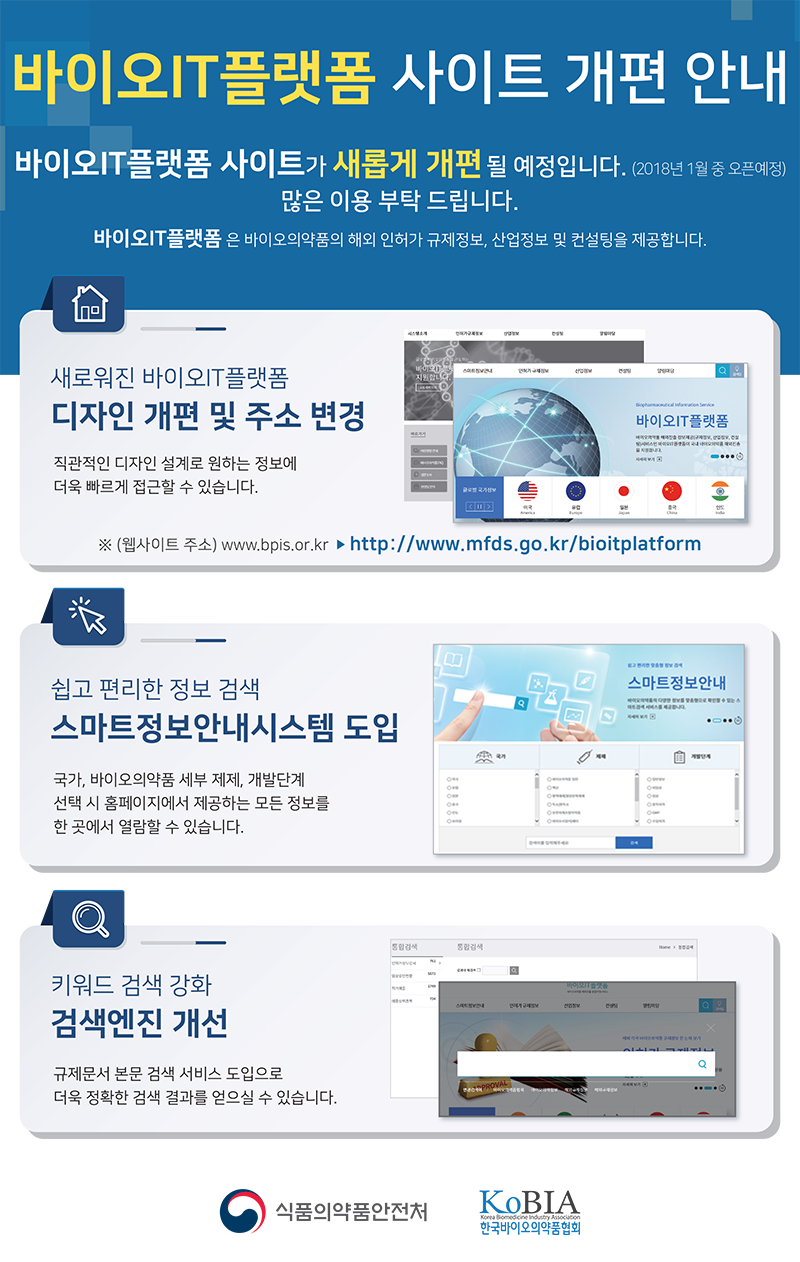 |
 |
| Ёс ЧљШИ МвНФ : ШИПјЛч ДыЛѓ '17Гт 12Пљ ЙпАЃ РкЗс ОШГЛ |
 |
 |
ЧбБЙЙйРЬПРРЧОрЧАЧљШИПЁМДТ ЁИ2017 ЙйРЬПРITЧУЗЇЦћЁЙ ЛчОїРЧ РЯШЏРИЗЮ
БЙГЛ ЙйРЬПРРЧОрЧАРЧ ЧиПм СјУтРЛ СіПјЧЯБт РЇЧи
Уб 15БЙПЁ ДыЧб БлЗЮЙњ СјУт РќЗЋ СЄКИ РкЗсС§РЛ ИЖЗУЧЯПДНРДЯДй.
(ЙЬБЙ/РЏЗД/РЯКЛ/СпБЙ/РЮЕЕ/КъЖѓСњ/ИпНУФк/ХЭХА/ХТБЙ/ШЃСж/ЗЏНУОЦ
/ЛчПьЕ№ОЦЖѓКёОЦ/КЃЦЎГВ/РЮЕЕГзНУОЦ/ЦФХАНКХК)
КЛ КИАэМПЁДТ АЂБЙ БдСІБтАќ СЄКИ, РЮЧуАЁ УМАш(ЁуРгЛѓНУЧш, ЁуЧАИёЧуАЁ, ЁуGMP), ЛъОїСЄКИ ЕюРЬ МіЗЯЕЧОю РжНРДЯДй. |
ЧбБЙЙйРЬПРРЧОрЧАЧљШИПЁМДТ ЙйРЬПРРЧОрЧА ЛъОїАш МКРх БтУЪ РкЗсИІ
СІАјЧЯАэРк 'ЙйРЬПРРЧОрЧА ЛъОїЕПЧт КИАэМ'ИІ ИЖЗУЧЯПДНРДЯДй.
КЛ КИАэМПЁДТ БЙГЛПм ЙйРЬПРРЧОрЧА НУРх ЧіШВРЬ МіЗЯЕЧОю РжНРДЯДй.
ЛѓММ ГЛПы: ШЈЦфРЬСі>РкЗсНЧ>ЧљШИАЃЧрЙА pdf ТќАэ
|
ЧљШИАЃЧрЙА АдНУЦЧ ЙйЗЮАЁБт [Click] |
ЛъОїЕПЧт КИАэМ ШЎРЮЧЯБт[Click] |
КИАэМ ПЖї МКёНКДТ ЧљШИ ШИПјЛч МвМг ШИПјРИЗЮ СІЧбЕЫДЯДй. ЧљШИ ШИПјЛч АЁРд Р§Тї ШЎРЮЧЯБт (Click) |
 |
| |
|
| |
|
ДКНКЗЙХЭ БИЕЖ НХУЛ: info@kobia.kr (ЙЋЗс)
(06725) МПяНУ МУЪБИ ГВКЮМјШЏЗЮ 333Бц 10, ПјРЯBD 4F
COPYRIGHT(C). ЧбБЙЙйРЬПРРЧОрЧАЧљШИ, All Rights Reserved.
|
 |
|
|
 > СЄКИИЖДч > E-ДКНКЗЙХЭ > РќУМ
> СЄКИИЖДч > E-ДКНКЗЙХЭ > РќУМ
 > СЄКИИЖДч > E-ДКНКЗЙХЭ > РќУМ
> СЄКИИЖДч > E-ДКНКЗЙХЭ > РќУМ